|
| Ribosome, mRNA, tRNA image by Sanbonmatsu Team, Los Alamos National Laboratory |
| |
|
|
|
| 3.1 Chemical elements and water |
Notes the Open Door web site
Notes Click4biology
|
|
|
|
3.1.1 State that the most frequently occurring chemical elements in living things are carbon, hydrogen, oxygen and nitrogen. |
Most common elements in living things: C, H, O, N |
|
|
|
3.1.2 State that a variety of other elements are needed by living organisms, including sulfur, calcium, phosphorus, iron and sodium. |
Other common elements: S, Ca, P, Fe, Na |
|
|
|
3.1.3 State one role for each of the elements mentioned in 3.1.2. |
Examples of functions:
|
|
|
|
3.1.4 Draw and label a diagram showing the structure of water molecules to show their polarity and hydrogen bond formation.
|
Diagram of Water Molecules showing polarity (+/-) and Hydrogen Bonding.
http://www.tutorvista.com/content/biology/biology-iii/cellular-micromolecules/water.php |
|
|
|
3.1.5 Outline the thermal, cohesive and solvent properties of water. |
Thermal Properties of Water
Cohesive Properties of Water
Solvent Properties of Water
Diagram of Ammonia dissolved in Water - Note the NH3+ molecules
http://www.chem.wisc.edu/deptfiles/genchem/sstutorial/Text7/Tx75/tx75.html
|
|
|
|
3.1.6 Explain the relationship between the properties of water and its uses in living organisms as a coolant, medium for metabolic reactions and transport medium. |
Water as a coolant in organisms:
Water as a medium for metabolic reactions:
Water as a transport medium:
|
| |
|
|
|
|
3.2 Carbohydrates, lipids and proteins |
|
|
|
|
3.2.1 Distinguish between organic and inorganic compounds. |
Organic compounds are based on carbon atoms and are found in living things. Some carbon compounds are not considered organic, such as CO2 and HCO3-
Inorganic compounds are all other compounds that are not organic compounds.
|
|
|
|
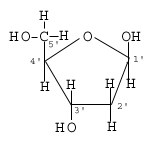
3.2.2 Identify amino acids, glucose, ribose and fatty acids from diagrams showing their structure. |
Monosaccharides (simple sugars) Glucose Ribose
Amino Acid
Fatty Acids
(Extra Information: All 20 Amino Acids)
http://www.ebi.ac.uk/2can/tutorials/aa.html
|
|
|
|
3.2.3 List three examples each of monosaccharides, disaccharides and polysaccharides. |
Monosaccharides
Disaccharides
Polysaccharides
|
|
|
|
3.2.4 State one function of glucose, lactose and glycogen in animals, and of fructose, sucrose and cellulose in plants. |
Animals
Plants
|
|
|
| 3.2.5
Outline the role of condensation and hydrolysis in the relationships between monosaccharides, disaccharides and polysaccharides; between fatty acids, glycerol and triglycerides; and between amino acids and polypeptides. |
Condensation Reactions
(a) Two molecules of glycine, side by side, showing how a water molecule can be formed using OH from one and H from the other. (b) The two glycine molecules are linked together to form a dipeptide. From: http://labspace.open.ac.uk/mod/resource/view.php?id=388779
Hydrolysis Reactions
|
|
|
| 3.2.6
State three functions of lipids. |
Lipid Functions
|
|
|
| 3.2.7
Compare the use of carbohydrates and lipids in energy storage. |
Comparison of Carbohydrates and Lipids Carbohydates provide about 4 Calories per gram. Lipids provide about 9 Calories per gram.
Carbohydrates are more easily broken down than lipids. Lipids require more oxygen to be broken down.
Carbohydrates are soluble in water, and lipids are not. Carbohydrates are easy to move around in solution, lipids are not.
|
 |
|
||
| 3.3 DNA structure |
Notes Click4biology
|
|
|
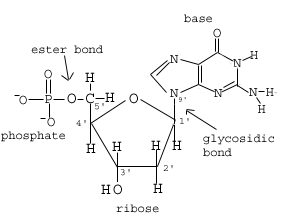
| 3.3.1
Outline DNA nucleotide structure in terms of sugar (deoxyribose), base and phosphate. |
A nucleotide consistes of three parts:
|
|
|
| 3.3.2
State the names of the four bases in DNA. |
Bases Adenine Thymine Cytosine Guanine
|
|
|
| 3.3.3
Outline how DNA nucleotides are linked together by covalent bonds into a single strand. |
Nucleotides are covalently bonded from the sugar of the first nucleotide to the phosphate of the second nucleotide.
A simple diagram of two nucleotides joined together:
Note: below is a lot more detail than you need to know.
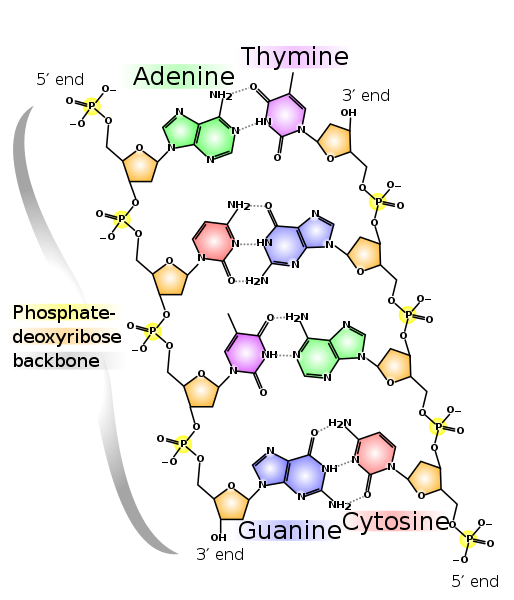
In a deoxyribose sugar the carbons are numbered from 1' to 5'
In a nucleotide, the phosphate is bonded to the 5' carbon, and the base is bonded to the 1' carbon.
The phosphate of the next nucleotide is covalently bonded to the 3' carbon.
The "backbone" of DNA is often referred to as a phosphate-sugar backbone going from 5' to 3'.
Note: when speaking, 5' is said as "five prime"
|
|
|
| 3.3.4
Explain how a DNA double helix is formed using complementary base pairing and hydrogen bonds. |
Hydrogen bonds form between pairs of bases:
The base pairs hydrogen bond in only this arrangement. The sizes of the bases and the hydrogen bonds will not fit together in any other arrangement.
This is called complementary base pairing.
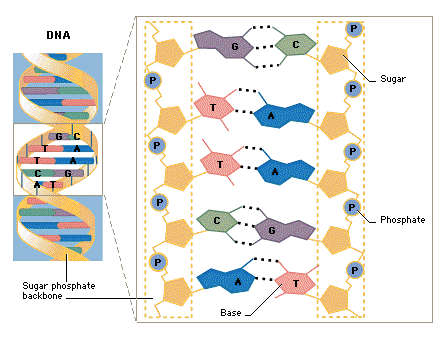
DNA has a double helix structure.
Sometimes it is called a "twisted ladder."
|
|
|
| 3.3.5
Draw and label a simple diagram of the molecular structure of DNA. |
|
| |
|
|
|
|
3.6 Enzymes
|
Notes Open Door
(includes HL topics)
Notes Click4Biology
|
|
|
| 3.6.1
Define enzyme and active site. |
An enzyme is a kind of protein that acts as a catalyst.
The active site is the place on the enzyme that holds the reactants.
|
|
|
| 3.6.2
Explain enzyme–substrate specificity. |
Enzyme-Substrate Specificity
The Lock and Key Model describes enzyme-substrate specificity.
|
|
|
| 3.6.3
Explain the effects of temperature, pH and substrate concentration on enzyme activity. 3.6.4 Define denaturation.
|
Effect of Temperature on Enzyme Activity
pH and Enzyme Activity
Enzyme Activity and Substrate Concentration
|
|
|
| 3.6.5
Explain the use of lactase in the production of lactose-free milk. |
Lactose-Free Milk
Lactose is a disaccharide made of a glucose and a galactose bonded together. It is known as milk sugar because it is found in the milk of mammals.
Lactase is an enzyme that breaks down lactose into glucose and galactose.
Many humans are lactose intolerant - this means they cannot digest lactose and feel sick when they consume milk products. Companies produce lactose-free milk for people who are lactose intolerant. The commercial production begins with lactase taken from yeast. The enzyme is attached to structural proteins so that it isn't lost in the process.
Milk is added to the beads and recirculated until all the lactose is broken down. The lactose-free milk is ready for the consumer. Enzymes are useful for this process and many others because:
See Click4Biology for a diagram of the industrial process.
|
| |
|
|
|
|
3.4 DNA replication |
http://genmed.yolasite.com/fundamentals-of-genetics.php
Notes Click4Biology
|
|
|
|
3.4.1 Explain DNA replication in terms of unwinding the double helix and separation of the strands by helicase, followed by formation of the new complementary strands by DNA polymerase. 3.4.2 Explain the significance of complementary base pairing in the conservation of the base sequence of DNA. 3.4.3 State that DNA replication is semi-conservative.
|
Watch the video and note these things:
1. Helicase 2. Replication fork 3. Leading strand 4. Lagging strand 5. DNA polymerase III 6. Free nucleotides 7. Complementary base pairing 8. Template 9. Semi-conservative replication 10. Result
|
|
|
| |
|
|
|
|
3.5 Transcription and translation |
Notes Click4Biology
|
|
|
|
3.5.1 Compare the structure of RNA and DNA.
http://skye-humanbio.blogspot.com/2008/09/unit-1-compendium-review.html |
DNA & RNA
Similarities
Differences
Uracil is very similar in shape and hydrogen bonding to thymine. So, the complementary RNA nucleotide for A is U.
A:::U |
|
|
| 3.5.2
Outline DNA transcription in terms of the formation of an RNA strand complementary to the DNA strand by RNA polymerase. |
Transcription: DNA --> RNA
Transcription
Three kinds of RNA
Ribosomes are structures in the cytosol of a cell and on the endoplasmic reticulum that build proteins.
|
|
|
| 3.5.3
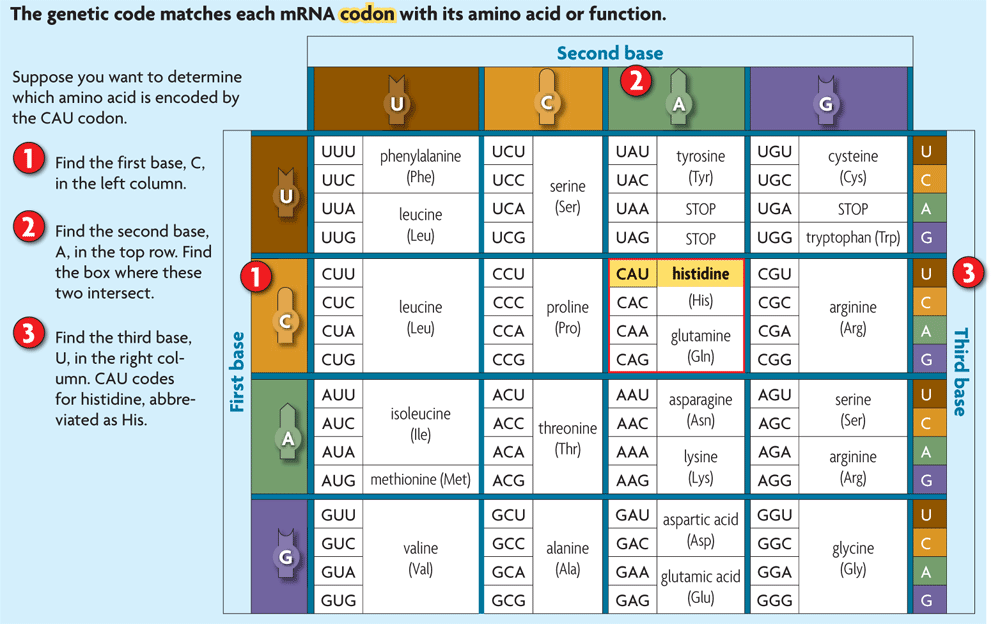
Describe the genetic code in terms of codons composed of triplets of bases. |
DNA is a book. A gene is a sentence. A nucleotide is a letter. What are the words? Triplets and Codons What do the words say? "Put this amino acid in the protien."
A triplet is a three nucleotide sequence.
A codon is a triplet in mRNA. The DNA triplet is transcribed into an mRNA codon. mRNA codons and their amino acids are identified in the genetic code.
In the genetic code below, the codon CAU is shown to code for the amino acid histidine.
|
|
|
| 3.5.4
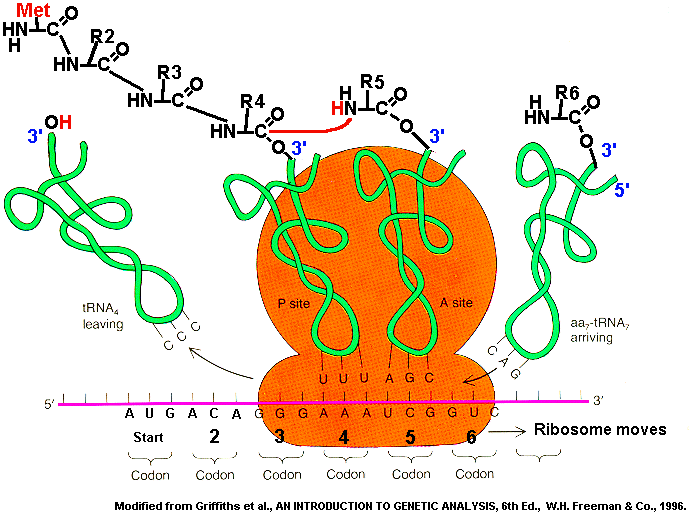
Explain the process of translation, leading to polypeptide formation. Include the roles of messenger RNA (mRNA), transfer RNA (tRNA), codons, anticodons, ribosomes and amino acids. |
Translation: RNA --> Proteins
How are the RNA codons translated into an amino acids in a protein?
Ribosomes use mRNA, tRNA, amino acids to build a new protein. mRNA is a copy of a gene. It has the sequence of amino acids for making a protein coded in its nucleotide sequence. tRNA has a triplet called an anticodon which is complementary to an mRNA codon. The tRNA has a specific amino acid bonded to the other end.
One mRNA codon matches one tRNA anticodon with one amino acid attached. The ribosome holds the mRNA and the tRNA together. Three mRNA codons fit inside the ribosome, and tRNAs can match their anticodons to the mRNA codons. The ribosome acts like an enzyme and bonds the amino acids on the adjacent tRNAs together. The ribosome moves down the mRNA and new tRNAs enter and their amino acids get bonded to the growing polypeptide. At the end of the mRNA, the ribosome falls apart and releases the mRNA, tRNAs, and the polypeptide (protein).
Transcription and Translation video
Transcription & Translation video from Shadowlabs
DNA Workshop - Interactive Shockwave Simulation
Transcription and Translation Model project
|
|
|
| 3.5.5
Discuss the relationship between one gene and one polypeptide.
|
Historical Progression of the One Gene/One Polypeptide Hypothesis 1. one gene makes one enzyme - discovered in the 1940s 2. one gene makes one protein - DNA makes other kinds of proteins 3. one gene makes one polypeptide - some proteins have multiple subunits Now 4. one gene makes multiple polypeptides - mRNA can be modified after transcription to make different polypeptides from te same gene.
|















![[dna-rna.gif]](http://1.bp.blogspot.com/_hWdXfsb45wQ/SNKf2c4bd6I/AAAAAAAAABE/vy7_bxT24ms/s1600/dna-rna.gif)